Activation of small molecules
The catalytic activation of small molecules is particularly relevant when thinking about CO, CO2, CH4, H2O, NOx etc that nowadays can be regarded as feedstock to obtain value added chemicals and/or energy, with the view to sustainable chemistry and cleaner production. The activation of these molecules is a challenging task in catalysis, as they are very stable and require active and selective catalysts to be converted to useful products or energy carriers. In our group we have been working for nearly three decades on ceria-containing materials that can exploit the unique redox properties of ceria to boost the activity and tune the selectivity of metal-based catalysts. The research is focused on the study of metal-support interactions that can modify catalytic properties and performances, trying to understand the fundamental relationship between structure and activity. This is done by in depth material characterization, coupling conventional and in situ techniques available in our lab and in collaboration with national and international partners.
Selected publications
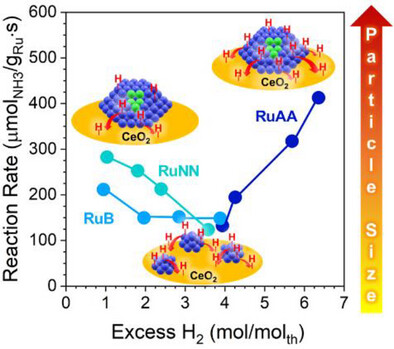
Parametric Study on Ru/CeO2 Ammonia Decomposition Catalysts for Hydrogen Production
A. Felli, M. Danielis, A. Trovarelli, and S. Colussi
ChemCatChem (2025) in press
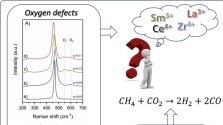
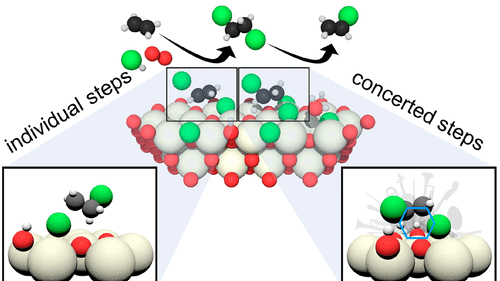
Low-Temperature Methane Activation Reaction Pathways over Mechanochemically-Generated Ce4+/Cu+ Interfacial Sites
S. Mauri, R. Calligaro, C. F. Pauletti, M. Farnesi Camellone, M. Boaro, L. Braglia, S. Fabris, S. Piccinin, P. Torelli, A. Trovarelli
Small 20 (2024) 2403028
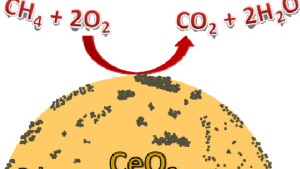
Investigation of the evolution of Pd-Pt supported on ceria for dry and wet methane oxidation
N. J. Divins, A. Braga, X. Vendrell, I. Serrano, X. Garcia, L. SOler, I. Lucentini, M. Danielis, A. Mussio, S. Colussi, I.J. Villar-Garcia, C. Escudero, A. Trovarelli, J. Llorca
Nature Commun., 13 (2022) 5080.
Methane oxidation activity and nanoscale characterization of Pd/CeO2 catalysts prepared by dry milling Pd acetate and ceria
M. Danielis, L.E. Betancourt, I. Orozco, N.J. Divins, J. Llorca, J.A. Rodríguez, S.D. Senanayake, S. Colussi, A. Trovarelli
Applied Catalysis B: Environmental, 282 (2021) 119567.
Structure-activity relationship in Pd/CeO2 methane oxidation catalysts
S. Colussi, P. Fornasiero, A. Trovarelli
Chinese J. Catal., 41 (2020) 938-950.
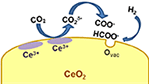
Ceria-Based Materials in Hydrogenation and Reforming Reactions for CO2 valorization
M. Boaro, S. Colussi, A. Trovarelli
Front. Chem., 7 (2019) 28.
Dry reforming of methane over Ni supported on doped CeO2: New insight on the role of dopants for CO2 activation
I. Luisetto, S. Tuti, C. Romano, M. Boaro, E. Di Bartolomeo, J.K. Kesavan,S. S. Kumar, K. Selvakumar
J. CO2 Utilization, 30 (2019) 63-78.
Mechanism of Ethylene Oxychlorination on Ceria
M. Scharfe, M. Capdevila-Cortada, V. A. Kondratenko, E.V. Kondratenko, S. Colussi, A. Trovarelli, N. López, J. Pérez-Ramírez
ACS Catal., 8 (2018) 2651-2663.